
And what about the one below? Is it right or wrong?: Algebra decides what will be the product and what will be the substrate! If you are an experienced chemist you will easily find the mistake in the mixed equation thanks to your chemical knowledge because the example is simple. But the calculator still works! Following the methodology described you will find that the value of some variables will be negative so they should be placed on the opposite side of the equation. If one did not notice the mistake one could not find the solution by means of the classical approach. What would happen if we mistakenly mixed it up a bit? To solve this system is to find the value of variables a, b, c which are the desired coefficients. With mathematics in mind you can write such a simple equation with variables a, b, c denoting the coefficients to be found:Īll you have to do is balance the quantity for each element on the left and the right side of the equation. Use the calculator with the explanation box checked to see the output. To see how the program finds the solution let`s start from a simple example.Īll the examples can be cut and pasted into the calculator 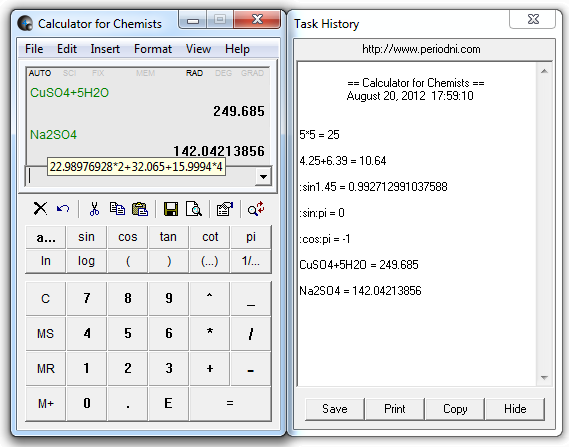
That students will find it very useful in determining the To prove that this algebraic technique works. It was a real shock when I realized the power of It provides better solutions than chemistry itself! SuchĪn idea might seem a bit strange for a chemist. Mathematics provides a general way to find reaction coefficients. Luckily it turns out that oxidation numbers and half-reaction are unnecessary! Another method exists that uses algebra to find the In some cases however it is hard to use this method because oxidation numbers cannot be unequivocally attributed. The classical approach uses oxidation numbers and many chemists cannot imagine any way to find the solution other than balancing half-reactions for the process. AsC l 3 aq + H 2 S aq → As 2 S 3 s + HCl aqĥ.Finding coefficients for chemical reactions is often complicated, especially in the case of redox processes. If no coefficients are required to balance the equation, simply place the integer 1 in front of each reactant and/or product.Ĭlick "Check" to see whether your response below is the correct balanced chemical equation.ģ. Type integer coefficients in front of the reactants and products to balance the chemical equations. In the following examples, you will be given the chemical equations and states. Identify the states of matter: use the following subscripts for the compounds: (g) for gaseous substances, (s) for solids, (l) for liquids, and (aq) for aqueous solutions. 
Finally, write out the products on the right hand side of the equation using chemical nomenclature.īalance the equation: use the Law of Conservation of Mass to balance, or achieve the same number of atoms of every element on each side of the equation. The direction of the arrow indicates the flow of the reaction. Draw an arrow to represent "forms to react". Remember to use uppercase and lowercase letters, where appropriate. 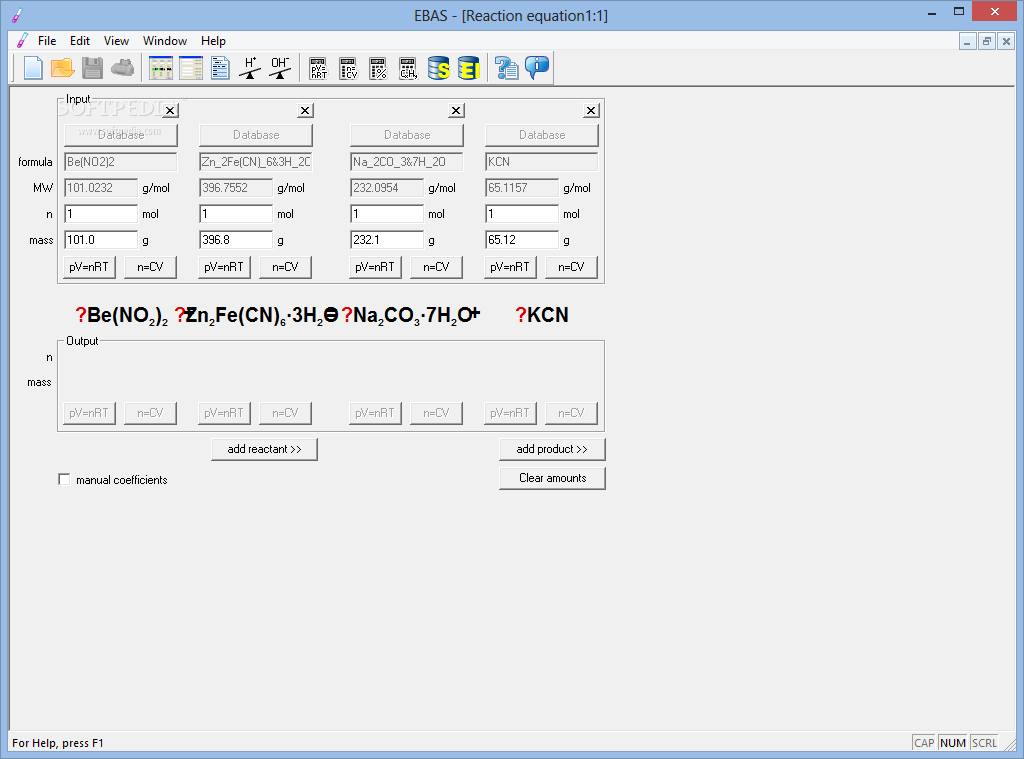
Write the unbalanced equation: using chemical nomenclature (see the Periodic Table of Elements ), write out the reactants on the left hand side of the equation. To balance chemical equations, the following three steps are used: Balancing a chemical equation is a mathematical approach used in chemistry to establish the relationship between the quantity of reactants and products. The equation shows the reactants on the left hand side and the products on the right hand side of the arrow. A chemical equation describes the changes that occur during a chemical reaction.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
